- Home
- Services
- About
- News
- Contact
- Logitech quickcam pro 5000 windows 10
- Universal audio 4110
- Arpspoof doesnt gather
- Getting risk 2 2 work on windows 10
- Sony vegas 16 video preview disappeared
- Lewis structure sf2
- Tom clancy the division pc how many players
- Magix music maker premium 2016
- Gta liberty city money cheats
- Change your airdrop name

Each atom will share, gain, or lose electrons to fill these outer electron shells with exactly eight electrons. Total valence e-: 20 e-F prefers 1 bond and follows the octet rule. According to the rule, atoms seek to have eight electrons in their outeror valenceelectron shells. Problem: Draw the Lewis structure for SF2.
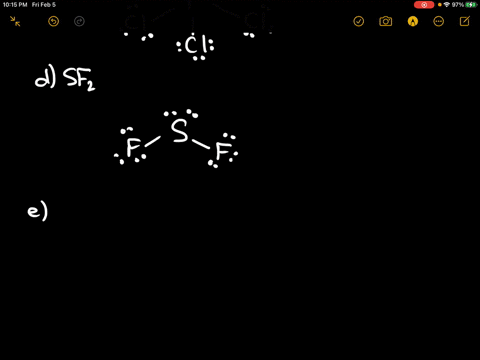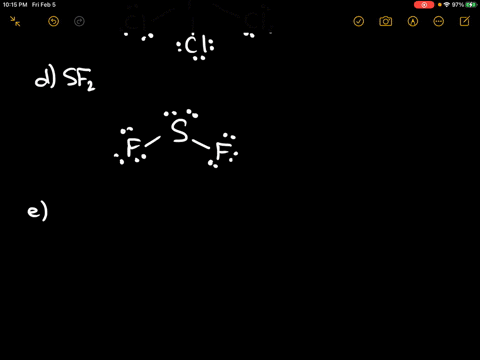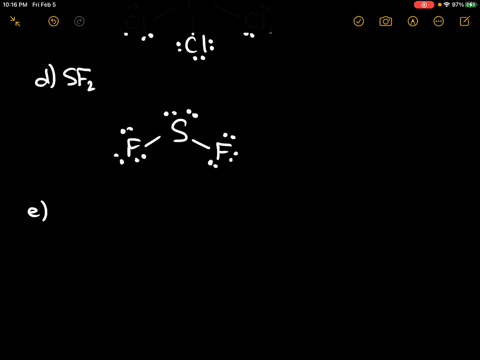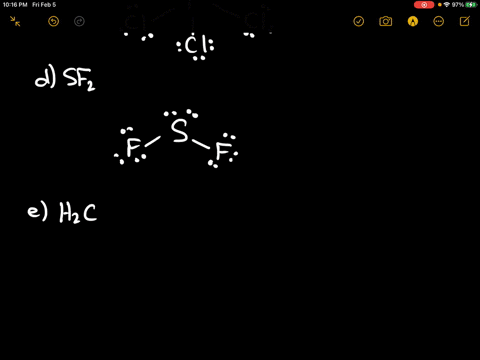
Even after bonding, the Beryllium atom does not contain 8 electrons in its valence shell. The octet rule is a bonding theory used to predict the molecular structure of covalently bonded molecules. The S is in the same family as O, but is larger than O, so the molecular shape is bent and the lone pairs are farther from the. Subsequently, one may also ask, what is the molecular shape of XeF2?īeryllium fluoride is linear because Beryllium only needs 4 electrons to attain an octet. So this is a possible lewis structure for sf2. The S is in the same family as O, but is larger than O, so the molecular shape is bent and the lone pairs are farther from the nucleus for S. Answered draw the lewis structure of sf2 bartleby molecular geometry deals with the overall shape of the molecule while electron geometry deals with the locations of the electrons around an atom. In the Lewis Structure of SF2, the central atom forms two bonds with two Fluorine atoms and has two lone pairs of electrons. The ideal angle for a tetrahedron is 109.5 deg.įurthermore, what is the bond angle of sf2? Answer: The bond angle on SF2 is not slightly less than 120 degrees, it is a lot less than 120 degrees. What is the Lewis structure of SF2 SF2 Shape. To calculate the formal charge on the central sulfur atom of the SF2 molecule by using the following formula: The formal charge on the sulfur atom of SF2 molecule (V. Thus, the orbitals would be in a tetrahedral arrangement and the molecule would be bent. SF2 would have a structure analogous to that of water - the central atom (S in SF2) has two lone pairs and two single bonds.
- Home
- Services
- About
- News
- Contact
- Logitech quickcam pro 5000 windows 10
- Universal audio 4110
- Arpspoof doesnt gather
- Getting risk 2 2 work on windows 10
- Sony vegas 16 video preview disappeared
- Lewis structure sf2
- Tom clancy the division pc how many players
- Magix music maker premium 2016
- Gta liberty city money cheats
- Change your airdrop name
